Selection of stainless steel for handling acetic acid CH3COOH
Acetic acid is a weak reducing acid. It is used in plastics manufacture and is a constituent of foods as vinegar. Ferritic stainless steels such as 430 type can be considered but normally the 304 types are used for most applications, including handling and storage.
Acetic anhydride (CH3CO)2O can be aggressive to either 304 or 316 types in the absence of any water and in the presence of chlorides. Peracetic acid CH3C(O)OOH (peroxyacetic acid) should be safe with stainless steel.Vinyl acetate C4H6O2 may be considered with the 316 grades for ambient temperature storage applications.
Commercially concentrated acid is around 99wt. % (glacial acetic acid).
Corrosion resistance of stainless steel
Ferritic stainless steel such as the 430 / 1.4016 type can be considered for most acid concentrations at ambient temperatures, but normally austenitic are preferred as pitting corrosion has been reported in industrial plant and equipment.
The 304 types are normally considered as suitable grades for most applications, including handling and storage.
The iso-corrosion diagram 0.1mm/year lines for the 316 / 1.4401 (red) types show that they can be expected to provide better resistance over about 5% concentrations, at temperature over 90 degC, than the 304 (blue) types.
Stainless Steel for Acetic acid, CH3COOH
| Conc. CH3COOH% |
1 |
1 |
5 |
5 |
5 |
5 |
10 |
10 |
10 |
20 |
| Temp. °C |
90 |
100
=BP |
20 |
50 |
75 |
100
=BP |
20 |
75 |
100
=BP |
20 |
| Grade or type of alloy: |
|
|
|
|
|
|
|
|
|
|
| Carbon steel |
1 |
2 |
1 |
2 |
2 |
2 |
1 |
2 |
2 |
2 |
| 13Cr |
0 |
1 |
1 |
2 |
2 |
2 |
1 |
2 |
2 |
1 |
| 304/304L |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
1 |
0 |
| TP316/316L |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| 18Cr13Ni3Mo/317L |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| 17Cr14Ni4Mo / 1.4439 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| 904L / N08904 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| Sanicro 28 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| 254 SMO |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| 654 SMO |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| SAF 2304 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| SAF 2205 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| SAF 2507 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| Titanium (CP Ti) |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
Symbol clarification
These corrosion tables use a number of symbols, having the following meanings:
| Symbol |
Description |
| 0 |
Corrosion rate less than 0.1 mm/year. The material is corrosion proof. |
| 1 |
Corrosion rate 0.1—1.0 mm/year. The material is not corrosion proof, but useful in certain cases. |
| 2 |
Corrosion rate over 1.0 mm/year. Serious corrosion. The material is not usable. |
| p, P |
Risk (severe risk) of pitting and crevice corrosion. |
| c, C |
Risk (Severe risk) of crevice corrosion. Used when there is a risk of localised corrosion only if crevices are present. Under more severe conditions, when there is also a risk of pitting corrosion, the symbols p or P are used instead. |
| s, S |
Risk (Severe risk) of stress corrosion cracking. |
| ig |
Risk of intergranular corrosion. |
| BP |
Boiling solution. |
| ND |
No data. (Used only where there are no actual data to estimate the risk of localised corrosion instead of p or s). |
(The boiling point corresponds to the red line for 316)
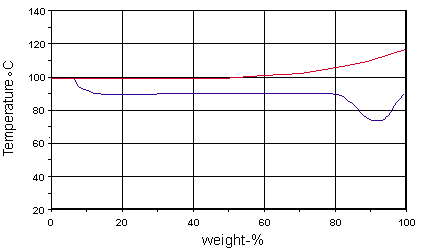 At concentrations above about 80%, the 316 types are usually considered a better choice than the 304 types, especially where temperatures exceed 70 degC, where there is a risk of localised attack to the 304 types.
For processing equipment, 316L is considered a better choice than the 304/304L or 316 types.
Intergranular attack can be an issue in weld heat affected zones, if acid contact temperatures exceed around 60 degC. As with other similar acid contact applications, the low carbon, 304L should be considered rather than the standard 304 types.
In common with most acid handling applications, chloride contamination can cause pitting corrosion and so in these cases more pitting resistant grades may need to be considered. Contamination of acetic acid with the more aggressive formic acid (HCOOH) can result in an unexpected reduction in corrosion resistance of the 316 types. The 304 types may be particularly vulnerable under these conditions.
Acetic anhydride (CH3CO)2O
Acetic anhydride (CH3CO)2O can be aggressive to either 304 or 316 types in the absence of any water and in the presence of chlorides.
The risk of pitting corrosion can be reduced if grades such as the austenitic 1.4539 or the 6% molybdenum grades are considered in these extreme conditions.
Peracetic acid CH3C(O)OOH
Peracetic acid CH3C(O)OOH which is also known as peroxyacetic acid, is used as a disinfectant (sanitiser) in food, medical and water treatment related industries.
It should be safe for uses that involve contact with stainless steel items.
Vinyl acetate C4H6O2
Vinyl acetate C4H6O2 is an intermediate product used in the manufacture of chemicals such as adhesives and paints.
The only information available suggests that the 316 types should be suitable for ambient temperature storage applications.
Releated References:
Selection of Stainless Steel fo Handling Sulphur Dioxide SO2 and Sulphur Trioxide SO3
Selection of Stainless Steel for Handling Phosphoric Acid H3PO4
Selection of Stainless Steel for Handling Hydrofluoric Acid HF
Selection of Stainless Steel for Handling Citric Acid C3H4OH (COOH)3
Selection of Stainless Steel for Handling Ammonia NH3
Selection of Stainless Steel for Handling Chlorine Cl2 and Chlorine Dioxide ClO2
Selection of Stainless Steels For Handling Hydrochloric Acid HCl
Selection of Stainless Steel for Handling Sulphuric Acid H2SO4
Selection Stainless Steel for Handling Sodium Hydroxide NaOH
Selection of stainless steels for handling acetic acid (CH3COOH)
Selection of stainless steels for handling sodium hypochlorite (NaOCl)
Selection of stainless steels for handling nitric acid (HNO3)
NACE MR 0175/ISO 15156 for Corrosion Resistant Alloys for Sulphide Service
Selection of stainless steels in water supply and waste water treatment
|
